Carbon dioxide extraction (Supercritical CO2)
Carbon dioxide (CO
2) extraction involves turning CO
2 from a gaseous state to a supercritical liquid state {call-out, text:A state between a liquid and a gas that is reached at a certain pressure and temperature. It can pass through solids like a gas and dissolve compounds like a liquid. Source:
Wikipedia}. This is done in two stages: first, by increasing the pressure (to 75 pounds per square inch), then by lowering the temperature of CO
2 (to -56.1°C, i.e. -69°F). Once those two parameters are met, a very slight increase in pressure and temperature is applied to make the CO
2 supercritical.
Using a super-critical extraction system, this super-critical liquid then passes through a pressure chamber holding the plant material and begins dissolving the trichome, thereby releasing the cannabinoids and terpenes into the CO
2. Once this process is finished, the liquid is then pulled through the machine to a separator that separates the CO
2 and other compounds from the desired cannabinoids and terpenes. Those desired compounds are then collected into a receptacle. The separated CO
2 can either be off-gassed into the air or recollected in another chamber on the machine to be reused.
1, 2
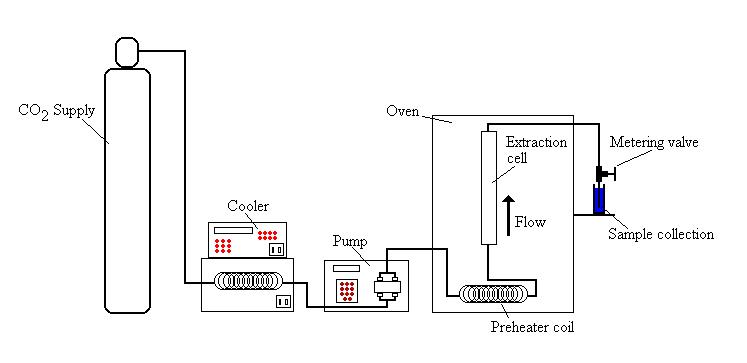 Figure 1: Schematic representation of superfluid extraction of cannabis compounds (image taken from Wikipedia).
Figure 1: Schematic representation of superfluid extraction of cannabis compounds (image taken from Wikipedia).
Watch a brief summary video on
Cannabis concentrates and extracts made in collaboration with
Enlighten on
YouTube or on
the screens page.
- Aladić, Krunoslav; Jarni, Kristjan; Barbir, Tina; Vidović, Senka; Vladić, Jelena; Bilić, Mate; Jokić, Stela (2015). Supercritical CO2 extraction of hemp (Cannabis sativa L.) seed oil. Industrial Crops and Products, 76, 472--478.
- Azmir, J.; Zaidul, I. S. M.; Rahman, M. M.; Sharif, K. M.; Mohamed, A.; Sahena, F.; Jahurul, M. H. A.; Ghafoor, K.; Norulaini, N. A. N.; Omar, A. K. M. (2013). Techniques for extraction of bioactive compounds from plant materials: A review. Journal of Food Engineering, 117(4), 426--436.
 Figure 1: Schematic representation of superfluid extraction of cannabis compounds (image taken from Wikipedia).
Watch a brief summary video on Cannabis concentrates and extracts made in collaboration with Enlighten on YouTube or on the screens page.
References:
Figure 1: Schematic representation of superfluid extraction of cannabis compounds (image taken from Wikipedia).
Watch a brief summary video on Cannabis concentrates and extracts made in collaboration with Enlighten on YouTube or on the screens page.
References:_logo.svg)